Chlorine gas has been used in the production of many solvents but it was not until 1915 during World War 1 that the german army used chlorine gas as a chemical weapon. It has a melting point of -155°c and a boiling point of -100°c. It is located in Group 17 as a non metal yellow gas with a pungent smell familiar with swimming pools at room temperature. It took many years before Humphrey Davy concluded that the new gas discovered was a new element, he named it Chlorine after the Greek ‘Chloros’ meaning yellow green. Chlorine was first produced by Carl Wilhelm Scheele in 1774 when he reacted Manganese dioxide with hydrochloric acid, at the time he did not know the name of these chemicals and instead named them as the alchemical forms of pyrolusite and spiritus salis. It is also used in the production of many plastics such as PVC and also the production of insecticides and solvents. 
It is used in swimming pools commonly and also making drinking water safe by killing bacteria. 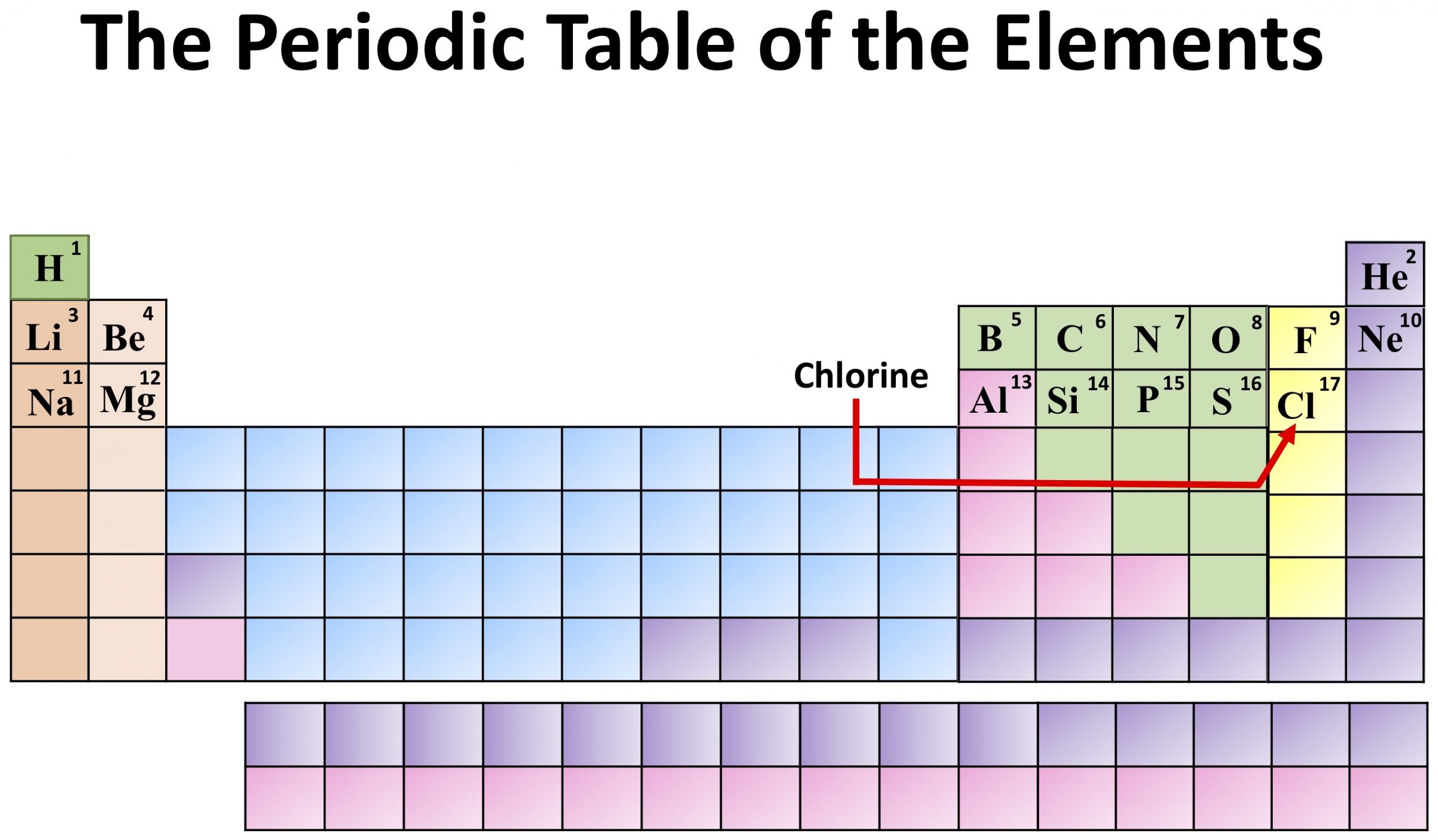
The van der Waals radius of chlorine is defined as half that distance or 184 pm. In solid chlorine the molecules pack together so that the shortest distance between chlorine nuclei in different molecules (AA or BB) or is 369 pm. It is a non metal with the symbol Cl.Ĭhlorine much like other Halogens has antiseptic properties. 2 The relationship between van der Waals radii and covalent radii for Cl 2 (s). It has the atomic number 17 in the periodic table and belongs in Group 17, the Halogens. When a very small cation combines with a very large anion, the resulting compound is less likely to exhibit the characteristic macroscopic properties of an ionic substance.Chlorine (Cl) exists as a yellow green gas with a pungent smell. The larger the anion, the farther it is from the sodium ion, and the weaker the coulombic force of attraction between them. The sizes of singly charged cations, for example, increase in the following order: Li + NaCl (808☌) > NaBr (750☌) > NaI (662☌). If we move down any of the columns, ionic sizes increase due to the increasing principal quantum number of the outermost electrons. Each row in the figure corresponds to an isoelectronic series involving a different noble-gas electron configuration.Īs we move from the more negative to the more positive ions in each row, there is a steady decrease in size. For any electronic series, such as H –, He, Li +, Be 2 +, in which the nuclear charge increases by 1 each time, we find a progressive decrease in size due to the increasingly strong attraction of the nucleus for the electron cloud. Species which have the same electronic structure but different charges are said to be isoelectronic. As we move across each row, the radius of each circle decreases. The final row has T e superscript 2 negative to T l superscript 3 positive. Fourth row has S e superscript 2 negative to I n superscript 3 positive. Third row has S superscript 2 negative to G a superscript 3 positive. The second row is N superscript 3 negative to Al superscript 3 positive. The first row is H negative to B e superscript 2 positive. Each row consists of elements as well as ions which have the same number of electrons. The elements of the periodic table are shown as circles with different radius. Ions in the first row of this figure, H –, Li +, and Be 2 +, all have the same 1 s 2 electronic structure as the helium (He) atom, but differ in size due to the different number of protons each has in their nucleus. This relationship is evident in the following figure comparing ionic radii. The size of an ion is governed not only by its electronic structure but also by its charge.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
